BYU professors developing spine disc replacement device
PROVO — Lower back pain plagues millions of Americans every day. In fact, back pain is the second leading reason people go to the doctor, after the common cold. But two BYU professors are working on relieving America’s back pain problems.
Mechanical engineers Larry Howell and Anton Bowden have developed a spine disc replacement device using flexible strips of titanium that move with the spine just like a natural disc.
The idea of spine disc replacement is nothing new, but attempts have not been very successful, Bowden said. Models on the market are typically made of metal and plastic like knee and hip replacement devices. The spine, however, functions differently from knees and hips, meaning different technology is needed.
“In your knee and hip those joints work because they have very low friction and that is what metal sliding on plastics is; it has very low friction and gives almost no resistance,” Bowden said. “But the spine needs to have some friction to work well.”
Currently there are only two options for relief from chronic lower back pain from a degenerating or ruptured disc — surgery that removes part of the ruptured material and spinal fusion. Fusion, Bowden said, causes more problems than it fixes.
Spinal fusion works by growing two sections of the spine together. A damaged disc is removed and the space is filled with bone fragments that eventually grow together. Howell said more than 300,000 spinal fusions are done in the U.S. each year, but the surgery limits spinal movement and causes wear and tear on other discs.
“Your spine needs to move to be healthy, so it is not uncommon for the discs next to the fusion to degenerate, and then patients have to go in and get another disc fused. It just cascades,” Howell said. “We hope that as we mimic the behavior of the spine with the artificial discs, it will help the discs nearby not have the same problems.”
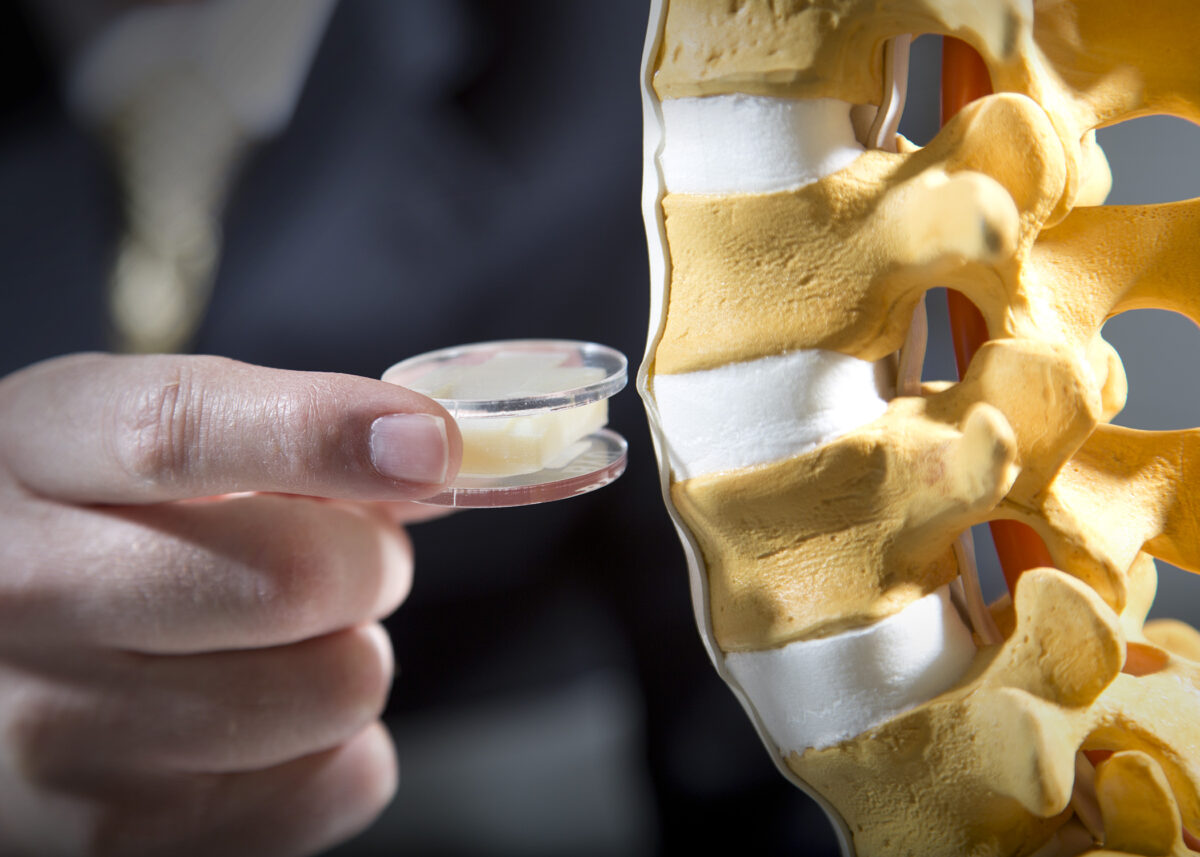
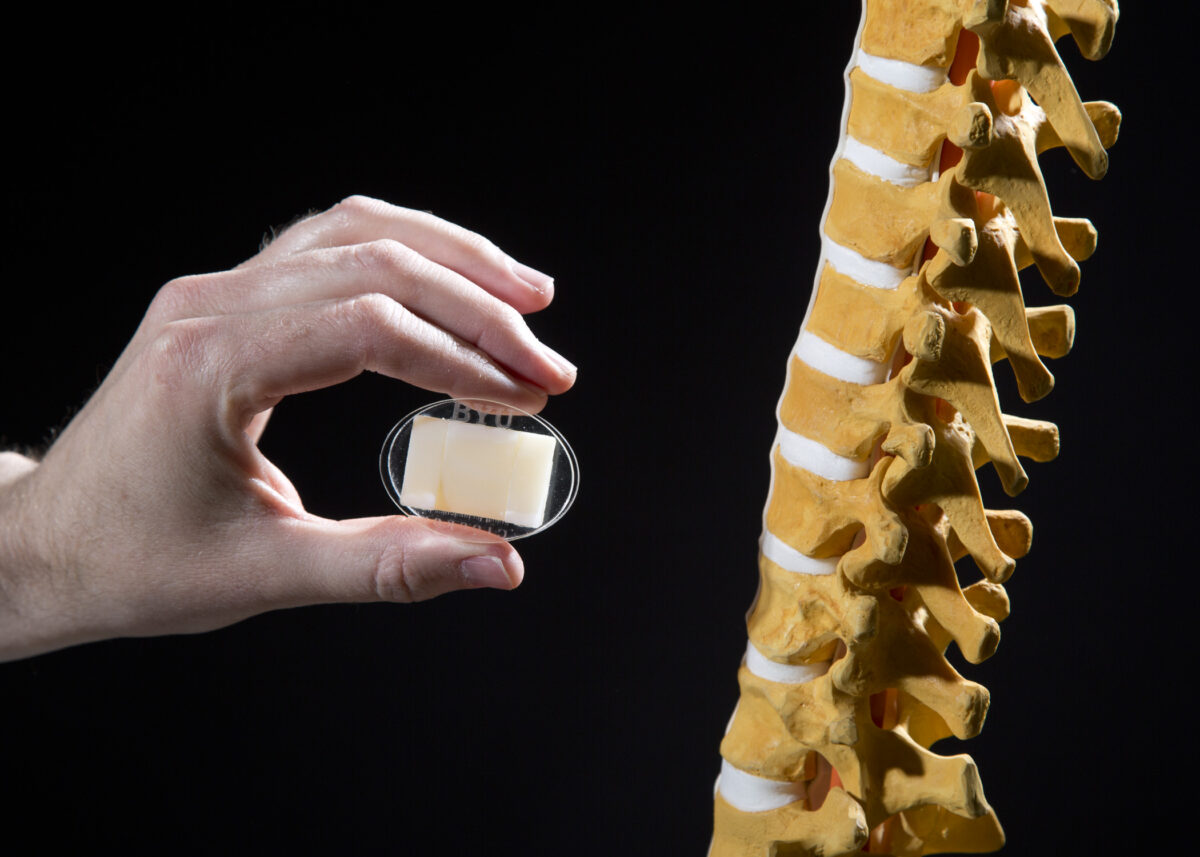
The disc that Howell and Bowden have designed, with the help of former doctoral student Peter Halverson, is made of strips of titanium or “bending beams” joined in such a way as to provide a flexing motion that mimics the natural movement of the spine, unlike hinge-like designs.
Titanium plates above and below the flexing strips have three fins that project into the adjoining vertibrae bones. The bone grows to these fins, thereby locking the flex mechanism in place. (See animation at artificialdisc.notlong.com)
“In order to make sure the device worked like we intended, we went through a pretty rigorous design process,” Bowden said. “We used computer models and then put it in cadaver spines and moved them like you move every day. We made sure that movement matched with how healthy spines move.”
The technology is being marketed by Salt Lake City-based Crocker Spinal Technologies and is set to hit the market overseas early next year.
David Hawkes, president of Crocker Spinal Technologies, said because of strict FDA regulations and the enormous cost of a clinical trial in the United States, they will be marketing overseas first.
“Because of the regulatory hurdles of the FDA, we will just start selling them internationally in 2013,” Hawkes said. “The FDA has said that if you have a well-controlled study that goes on outside the U.S. you can bring that data back and show that it worked well outside the U.S. but they have never actually allowed that, they always say you will have to re-create the study in the U.S.”
Hawkes said for now they will market overseas in hopes the FDA will lower its regulations or accept their data after a few years of performing the disc replacement. If that doesn’t work, they will eventually look into doing a trial in the U.S. He said right now the latest and greatest technologies are only available internationally due to strict FDA regulations.
Howell, Bowden and Hawkes agree that regardless of how long it takes for the technology to be available here, it has the potential to change people’s lives.
“One of the key things is the potential this has to really help improve the quality of life for people who suffer from low back pain, and that is just an exciting prospect,” Howell said. “It is also exciting to use cutting edge technology to do something that has never been done before and that will be useful in the world.” Hawkes said the disc that hits the market early next year will be a model used for cervical spine (neck area) procedures and the lumbar model will follow with in 18 months to two years.